 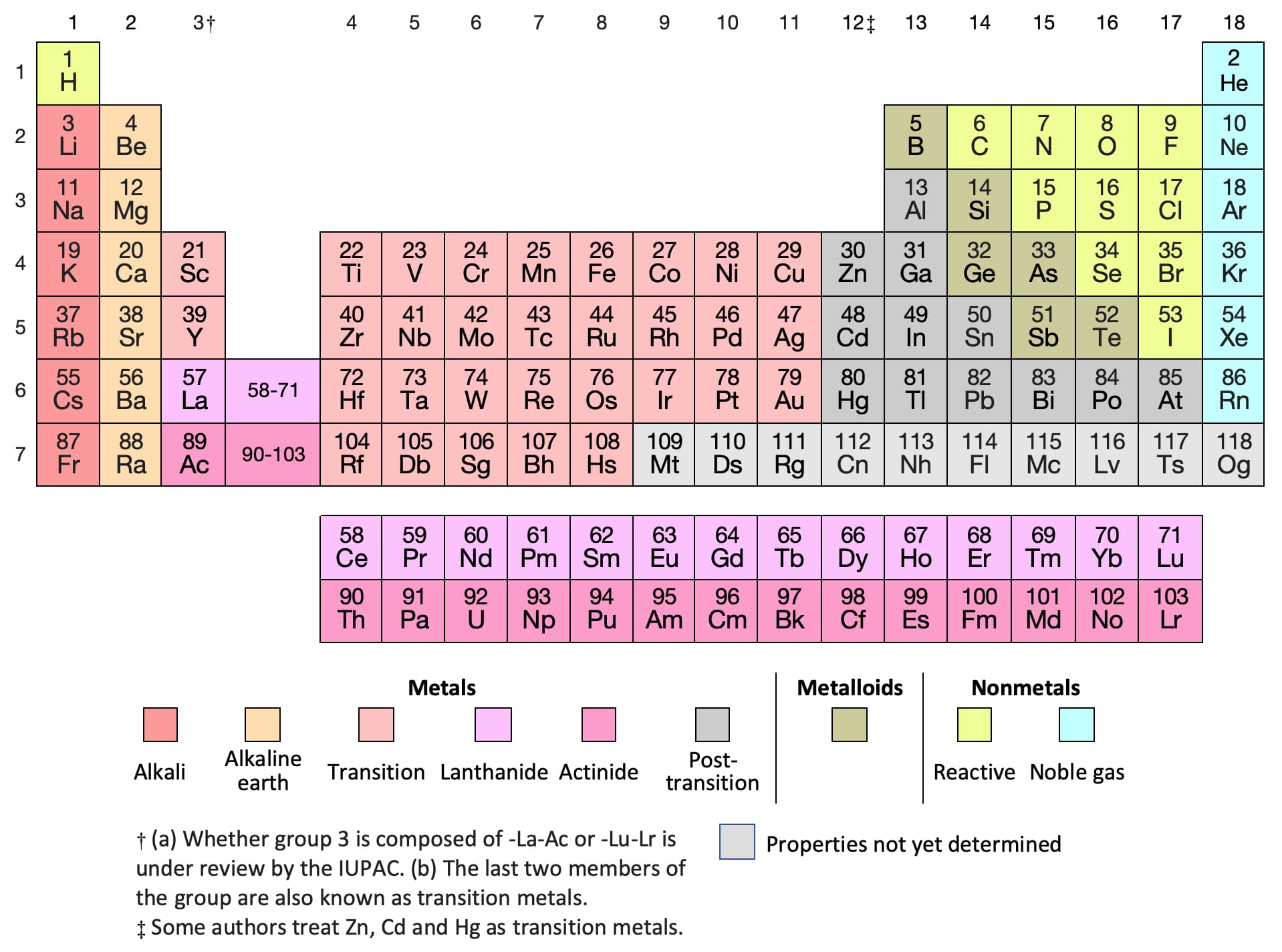 
A larger number indicates a larger and higher energy orbital. This number is an indication of the size and energy of the orbital. Orbitals are often preceded by numerical designations, i.e. These elements use electrons in the d orbitals for bonding and chemical reactivity. Of d orbitals are called transition, or d-block, elements. 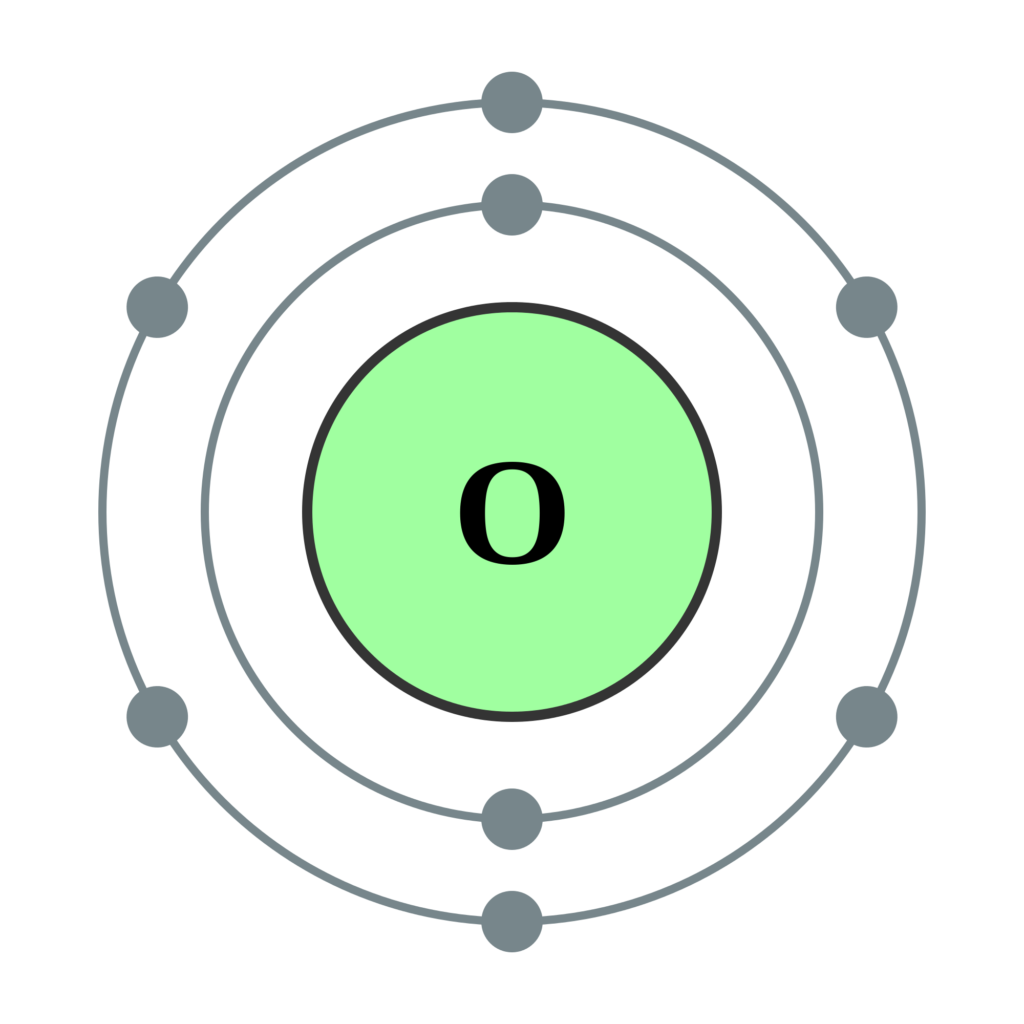
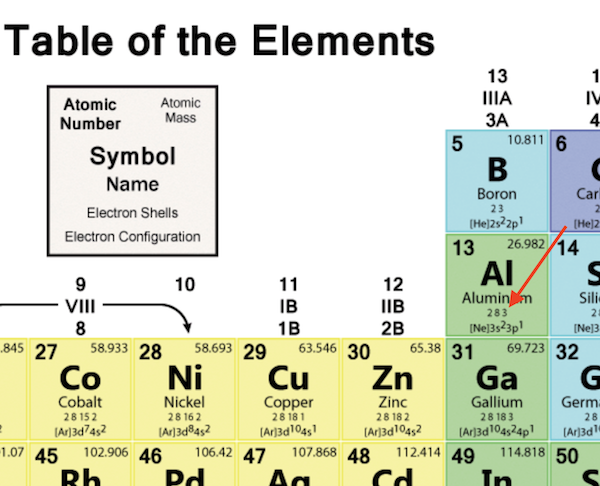
For example, elements having a partially filled set The type of orbital (s, p, d, or f) that the valence electrons reside in is a function of the elements' position in the periodic table. These electrons, called the valence electrons, are the most loosely held and interact with those in other atoms to form chemical bonds. The chemical and physical behavior of the elements results from the configuration of the outermost electrons. The shape and orientation of the d orbitals, which together can hold up to 10 electrons, are shown to the right. There are five d orbitals, which have more complicated shapes than s and p orbitals. The p orbitals can hold up to six electrons. There are three p orbitals, each of which has the same basic dumbbell shape but differ in its orientation in space. An s orbital has a spherical shape and can hold two electrons. There are four basic types of orbitals: s, p, d, and f. An orbital is a region of space where there is a high probability of finding an electron. Early models of the atom depicted the electrons circling the nucleus in fixed orbits, much like planets revolving around the sun.Ĭurrent theory suggests that electrons are housed in orbitals. Electrons are very light, negatively charged particles that surround the positively charged nucleus. The nucleus is the central core of an atom and is made up of protons and neutrons. In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is.The Actinide Research Quarterly: 1st Quarter 2004 contentsĪn atom consists of two basic parts: the nucleus and the electrons. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. For non-transition metals, we count to 8, but for transition metals, we count to 12. Looking at the orbitals explains how valence electrons work for transition metals. For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). The shells after (ignoring transition metals) represent the s and p-orbitals. So what does this have to do with our shells? The first "shell" represents the 1s orbital. For example, carbon has an electron configuration of 1s 22s 22p 2. The way we count our electrons is by moving from right to left, starting at the beginning of the table. F-orbitals start appearing in the lanthanides and actinides (the separated two rows). P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level.Has 5 subshells, each holding 2 electrons, for a total of 10 electrons.Has 3 subshells, each holding 2 electrons, for a total of 6 electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
